NCERT Solutions for Class 11 Chemistry Chapter 4 – Chemical Bonding and Molecular Structure
📘 Get NCERT Solutions for Class 11 Chemistry Chapter 4 – Chemical Bonding and Molecular Structure with step-by-step explanations, solved NCERT questions, and practice MCQs. These solutions are prepared according to the latest CBSE syllabus and are helpful for Class 11 Board Exams, NEET, JEE and other competitive exams. Important topics covered in this chapter include Kossel–Lewis Approach, Ionic Bond, Covalent Bond, VSEPR Theory, Valence Bond Theory, Hybridization and Molecular Geometry. All concepts are explained in simple language so students can easily understand the formation and structure of molecules. ✅
To find any question quickly, use the Search Box and type either the Question Number (for example Q.10, Q.25) or a keyword from the question such as ionic bond, covalent bond, hybridization, VSEPR theory, or molecular geometry. ✅
- Example: Type Q.15 or hybridization in the search box.
- Tip: Using two or three keywords together will help you find results faster.
- Mobile Users: You can also use your browser option Find in Page (Chrome → Menu → Find in Page).
✅ The attractive force that holds atoms together in a molecule is called a chemical bond.
✅ According to the Kossel–Lewis theory, chemical bonds are formed mainly by:
1️⃣ Transfer of valence electrons
2️⃣ Sharing of valence electrons
✅ Through these processes, atoms try to achieve a stable octet electronic configuration.
👉 In most substances, atoms do not remain alone but combine to form stable groups called molecules.
👉 The force that holds these atoms together is known as a chemical bond.
Step 2: Definition of a chemical bond
A chemical bond is the attractive force that holds atoms or ions together in a chemical species.
Step 3: Kossel–Lewis concept
👉 Atoms form bonds in order to achieve greater stability.
👉 This stability is usually obtained by achieving the octet configuration of noble gases.
👉 This can occur either by:
• transfer of electrons
• sharing of electrons
Step 4: Example (Formation of NaCl)
👉 Sodium (Na) loses one electron to form Na+.
👉 Chlorine (Cl) gains one electron to form Cl−.
👉 The electrostatic attraction between Na+ and Cl− forms an ionic (electrovalent) bond.
👉 The formation of chemical bonds usually results in lower energy and greater stability.
👉 When electrons are transferred, an ionic bond is formed, and when electrons are shared, a covalent bond is formed.
• Mg (2,8,2) → 2 valence electrons
• Na (2,8,1) → 1 valence electron
• B (2,3) → 3 valence electrons
• O (2,6) → 6 valence electrons
• N (2,5) → 5 valence electrons
• Br (2,8,18,7) → 7 valence electrons
Lewis Dot Symbols:
• Mg : •Mg•
• Na : Na•
• B : •B••
• O : :O:•• (total 6 dots)
• N : :N••• (total 5 dots)
• Br : :Br:••• (total 7 dots)
Note: The positions of dots (top, bottom, left, or right) may vary, but the total number of dots must equal the number of valence electrons.
👉 A Lewis dot symbol represents the valence electrons of an atom by dots placed around its chemical symbol.
Step 2: How to determine Lewis dot symbols?
1️⃣ Write the electronic configuration of the element.
2️⃣ Identify the electrons in the outermost shell (valence electrons).
3️⃣ Represent these electrons as dots around the element symbol.
Step 3: Applying to the given elements
• Mg (12): 2,8,2 → 2 valence electrons
• Na (11): 2,8,1 → 1 valence electron
• B (5): 2,3 → 3 valence electrons
• O (8): 2,6 → 6 valence electrons
• N (7): 2,5 → 5 valence electrons
• Br (35): 2,8,18,7 → 7 valence electrons
👉 Lewis dot symbols are extremely useful for understanding chemical bonding, especially ionic bonds and covalent bonds.
👉 Elements like oxygen (6 valence electrons) and bromine (7 valence electrons) tend to gain or share electrons to achieve a stable octet configuration.
• S : :S:•• (6 valence electrons)
• S2− : [:S:]2− (8 electrons, octet complete)
• Al : Al••• (3 valence electrons)
• Al3+ : [Al]3+ (no valence electrons shown)
• H : H• (1 valence electron)
• H− : [:H:]− (2 electrons, duplet complete)
👉 Lewis symbols represent the valence electrons of an atom using dots around the element symbol.
Step 2: Determine valence electrons
• S → Group 16 → 6 valence electrons
• Al → Group 13 → 3 valence electrons
• H → Group 1 → 1 valence electron
Step 3: Effect of ion formation
👉 When atoms gain or lose electrons, their Lewis symbols change.
• S2− gains 2 electrons → total 8 electrons (octet).
• Al3+ loses 3 electrons → no valence electrons shown.
• H− gains 1 electron → 2 electrons (duplet configuration).
👉 Most atoms follow the octet rule, while hydrogen follows the duplet rule.
👉 Lewis symbols are extremely useful for understanding ionic and covalent bonding.
1️⃣ H2S
H – S – H
(Sulfur has two lone pairs)
2️⃣ SiCl4
Cl – Si – Cl
| Cl Cl
(Si forms four single bonds with Cl atoms)
3️⃣ BeF2
F – Be – F
(Linear molecule; Be forms two bonds)
4️⃣ CO32−
O – C = O | O−
(Resonance occurs among three equivalent structures)
5️⃣ HCOOH (Formic Acid)
H – C = O | O – H
👉 Determine the total number of valence electrons of all atoms.
Example:
• H2S → S = 6, H = 1×2 → total = 8 electrons
• CO32− → C = 4, O = 6×3 + 2 extra electrons → total = 24 electrons
Step 2: Choose the central atom
👉 Usually the least electronegative atom becomes the central atom.
Example:
S in H2S, Si in SiCl4, C in CO32−.
Step 3: Form single bonds
👉 Connect surrounding atoms with single bonds first.
Step 4: Complete octet
👉 Add lone pairs to complete the octet rule.
👉 In some molecules like BeF2, the central atom may have incomplete octet.
Step 5: Consider resonance
👉 Some molecules like CO32− show resonance structures.
👉 CO32− shows three resonance structures, making all C–O bonds equivalent.
👉 Lewis structures help in predicting molecular shape and bonding.
✅ This rule was proposed by Kossel and Lewis (1916) based on the stability of noble gases.
✅ According to this concept, chemical bonds are formed mainly by:
1️⃣ transfer of electrons (ionic bond)
2️⃣ sharing of electrons (covalent bond)
👉 Noble gases are highly stable because their outermost shell is completely filled.
👉 Helium has 2 electrons (duplet), while most other noble gases have 8 electrons (octet) in the outer shell.
👉 Other atoms form chemical bonds to achieve this stable configuration.
Step 2: How is the octet achieved?
(a) By transfer of electrons
Example: Formation of sodium chloride
Na → Na+ + e−
Cl + e− → Cl−
Na+ + Cl− → NaCl
👉 Both atoms achieve a noble gas configuration.
(b) By sharing of electrons
👉 Two atoms share one or more pairs of electrons to obtain stable octet configuration, as seen in many covalent molecules.
Step 3: Significance of the octet rule
✅ It explains the formation of many molecules such as O2, N2, Cl2, Br2.
✅ It helps in understanding ionic and covalent bonding.
✅ It is useful in explaining the structure of many organic compounds.
✅ It works particularly well for second period elements.
Step 4: Limitations of the octet rule
The octet rule is not universally applicable. Important exceptions include:
1) Incomplete Octet
👉 In some compounds, the central atom has fewer than 8 electrons.
Examples: BeH2, BCl3, AlCl3
2) Odd Electron Molecules
👉 Some molecules contain an odd number of electrons, so the octet cannot be completed for all atoms.
Examples: NO, NO2
3) Expanded Octet
👉 Elements of the third period and beyond can have more than 8 electrons in their valence shell.
Examples: PF5, SF6, H2SO4
👉 Helium is stable with only two electrons, so it follows the duplet rule instead of the octet rule.
👉 In exams, when writing limitations of the octet rule, always mention these three main exceptions:
• Incomplete octet
• Odd electron molecules
• Expanded octet
✅ Low ionisation enthalpy of the atom forming the cation.
✅ High (more negative) electron gain enthalpy of the atom forming the anion.
✅ High lattice energy of the resulting ionic crystal.
These factors together increase the stability of ionic compounds.
👉 In an ionic bond, one atom loses an electron to form a cation.
👉 If the ionisation enthalpy is low, the electron can be removed easily.
👉 Therefore, atoms with low ionisation enthalpy favour the formation of ionic bonds.
Example: alkali metals and alkaline earth metals easily form cations.
Step 2: Effect of electron gain enthalpy
👉 The second atom gains an electron to form an anion.
👉 If the electron gain enthalpy is highly negative, the atom can accept electrons easily.
👉 This makes the formation of an ionic bond more favourable.
Example: halogens (Group 17) readily gain electrons.
Step 3: Effect of lattice energy
👉 When gaseous ions combine to form a solid ionic crystal, energy is released.
👉 This released energy is called lattice energy.
👉 Greater lattice energy means stronger ionic bonding and greater stability.
A+(g) + B−(g) → A+B−(s) + U
Here U represents lattice energy.
Step 4: Overall conclusion
👉 Ionic compounds are most stable when:
• Ionisation enthalpy is low
• Electron gain enthalpy is highly negative
• Lattice energy is large
This means the overall process should be energetically favourable.
👉 Ions with smaller size and higher charge attract each other more strongly, leading to greater lattice energy.
👉 Easy exam trick to remember favourable factors:
Low IE + Highly negative EGE + High Lattice Energy → Ionic bond formation
Molecular Shapes:
• BeCl2 → Linear (Bond angle ≈ 180°)
• BCl3 → Trigonal planar (Bond angle ≈ 120°)
• SiCl4 → Tetrahedral (Bond angle ≈ 109.5°)
• AsF5 → Trigonal bipyramidal (Bond angles ≈ 90° and 120°)
• H2S → Bent or V-shaped (Bond angle ≈ 92°)
• PH3 → Trigonal pyramidal (Bond angle ≈ 93.5°)
👉 According to the Valence Shell Electron Pair Repulsion (VSEPR) theory, electron pairs around a central atom arrange themselves as far apart as possible to minimize repulsion.
Step 2: Shapes of the given molecules
BeCl2
👉 Central atom Be has 2 bonding pairs and no lone pairs.
👉 Geometry → Linear (180°).
BCl3
👉 B has 3 bonding pairs and no lone pairs.
👉 Geometry → Trigonal planar (120°).
SiCl4
👉 Si has 4 bonding pairs.
👉 Geometry → Tetrahedral (109.5°).
AsF5
👉 As has 5 bonding pairs.
👉 Geometry → Trigonal bipyramidal.
H2S
👉 S has 2 bonding pairs and 2 lone pairs.
👉 Geometry → Bent or V-shaped.
PH3
👉 P has 3 bonding pairs and 1 lone pair.
👉 Geometry → Trigonal pyramidal.
👉 Example order of bond angles:
CH4 (109.5°) > NH3 (107°) > H2O (104.5°)
👉 VSEPR theory is widely used to predict molecular geometry and bond angles.
✅ In NH3, nitrogen has one lone pair.
✅ In H2O, oxygen has two lone pairs.
✅ Since lone pair–lone pair and lone pair–bond pair repulsions are stronger, the bond pairs in water are pushed closer together.
Therefore:
• NH3 bond angle ≈ 107°
• H2O bond angle ≈ 104.5°
👉 According to the VSEPR theory, electron pairs around the central atom repel each other and arrange themselves to minimize repulsion.
The order of repulsion is:
lone pair – lone pair > lone pair – bond pair > bond pair – bond pair
Step 2: Structure of NH3
👉 In ammonia, the central atom N has 4 electron pair regions:
• 3 bond pairs (N–H)
• 1 lone pair
👉 Because of lone pair–bond pair repulsion, the bond angle decreases from the ideal 109.5° to about 107°.
Step 3: Structure of H2O
👉 In water, the central atom O has 4 electron pair regions:
• 2 bond pairs (O–H)
• 2 lone pairs
👉 Because there are two lone pairs, repulsion is greater than in NH3.
👉 This pushes the O–H bond pairs closer together and reduces the bond angle to about 104.5°.
Step 4: Conclusion
👉 Although both molecules have tetrahedral electron geometry, the greater number of lone pairs in H2O increases repulsion and decreases the bond angle compared with NH3.
👉 Lone pairs occupy more space than bond pairs, so they push bonding atoms closer together.
👉 This gives the common bond angle order:
CH4 (109.5°) > NH3 (107°) > H2O (104.5°)
✅ This means that as the bond order increases, the bond becomes stronger.
✅ This happens because higher bond order usually results in greater bond dissociation enthalpy.
Bond Strength ∝ Bond Order
👉 The strength of a bond depends on the energy required to break the bond.
👉 This energy is known as bond dissociation enthalpy.
👉 The greater this energy, the stronger the bond.
Step 2: Effect of bond order
👉 Bond order represents the number of shared electron pairs between two atoms.
👉 When bond order increases, the attraction between atoms increases.
👉 As a result, the bond becomes shorter and stronger.
Step 3: Explanation with examples
• N2 : Bond order = 3, Bond enthalpy ≈ 945 kJ mol−1
• O2 : Bond order = 2, Bond enthalpy ≈ 498 kJ mol−1
👉 Since N2 has a higher bond order, its bond is stronger than that of O2.
Step 4: Conclusion
👉 Therefore, bond strength increases with increase in bond order:
Bond Order ↑ → Bond Strength ↑ → Bond Dissociation Enthalpy ↑
Single bond < Double bond < Triple bond
👉 As bond order increases, the bond length usually decreases.
👉 That is why molecules with triple bonds such as N2 are extremely stable and strong.
✅ It is usually expressed in picometre (pm).
1 pm = 10−12 m
👉 When two atoms form a chemical bond, a certain stable distance is established between their nuclei.
👉 This stable or equilibrium distance is called the bond length.
Step 2: Unit of bond length
👉 Bond length is extremely small, so it is generally measured in picometres (pm).
Step 3: In ionic compounds
👉 In ionic compounds, bond length is approximately equal to:
Radius of cation + Radius of anion
Step 4: In covalent compounds
👉 In covalent molecules, bond length is approximately equal to:
Sum of the covalent radii of the two bonded atoms
👉 A shorter bond usually indicates a stronger bond.
👉 Even between the same atoms, different types of bonds have different lengths:
Single bond > Double bond > Triple bond
✅ Instead, it is described by three equivalent resonance structures in which the double bond between carbon and oxygen is distributed among the three oxygen atoms.
✅ The actual structure is a resonance hybrid, and all C–O bonds are identical.
👉 In one Lewis structure, carbon forms a double bond with one oxygen and single bonds with the other two oxygen atoms.
👉 However, the double bond can be placed with any of the three oxygen atoms, giving three equivalent structures.
Step 2: Resonance concept
👉 The real structure of the carbonate ion is not any one of these structures but a resonance hybrid of all three.
👉 In this hybrid structure, the electrons are delocalized over all three C–O bonds.
Step 3: Consequences of resonance
👉 All three C–O bonds have the same bond length.
👉 The bond order of each C–O bond is 1⅓.
👉 The ion is more stable than any individual resonance structure.
👉 The extra stability obtained due to electron delocalization is called resonance stabilization.
👉 Many important molecules such as benzene and nitrate ion also show resonance.
👉 In resonance structures, only electrons shift while the positions of atoms remain the same.
👉 In the given structures, the hydrogen atom changes its position from oxygen to phosphorus.
👉 Therefore these two structures represent different compounds, not resonance forms.
👉 Resonance occurs when two or more structures differ only in the distribution of electrons.
👉 The positions of atoms must remain unchanged.
Step 2: Analysis of the given structures
👉 In structure (1), hydrogen is attached to phosphorus.
👉 In structure (2), hydrogen is attached to oxygen.
👉 This means the position of hydrogen changes.
Step 3: Correct structure of H3PO3
👉 The correct structure contains:
• One P=O double bond
• Two P–OH groups
• One P–H bond
👉 Therefore hydrogen is directly bonded to phosphorus in the actual molecule.
👉 It is actually diprotic, not triprotic, because only two hydrogens are attached to oxygen and can be ionized.
👉 The hydrogen attached to phosphorus (P–H) does not show acidic behavior.
SO3, NO2 and NO3−
1. SO3
👉 Sulphur forms three equivalent resonance structures.
O=S–O ↔ O–S=O ↔ O–S–O
(The double bond shifts among the three oxygen atoms.)
2. NO2
👉 Nitrogen dioxide has two resonance structures.
O=N–O ↔ O–N=O
(The double bond alternates between the two oxygen atoms.)
3. NO3−
👉 Nitrate ion has three resonance structures.
O=N–O⁻ ↔ O⁻–N=O ↔ O–N=O⁻
(The double bond moves among the three oxygen atoms.)
👉 Some molecules cannot be represented by a single Lewis structure.
👉 Instead, they are described by multiple structures called resonance structures.
Step 2: Resonance in SO3
👉 Sulphur is bonded to three oxygen atoms.
👉 The S=O double bond can appear with any oxygen atom.
👉 Therefore three equivalent resonance structures exist.
👉 As a result, all S–O bonds become identical.
Step 3: Resonance in NO2
👉 Nitrogen forms one double bond and one single bond with oxygen atoms.
👉 The double bond shifts between the two oxygen atoms.
👉 Hence two resonance structures are possible.
Step 4: Resonance in NO3−
👉 Nitrate ion contains three oxygen atoms bonded to nitrogen.
👉 One N=O double bond and two N–O single bonds are present in each structure.
👉 The double bond can shift among the three oxygen atoms.
👉 Therefore three equivalent resonance structures are formed.
👉 Because of resonance, all N–O bond lengths become equal.
👉 Resonance increases the stability of molecules because electrons become delocalized.
👉 In NO3−, all N–O bonds have the same bond length due to resonance.
(b) Ca and O
(c) Al and N
K → K+ + e−
S + 2e− → S2−
Lewis representation:
2K• + :S:•••• → 2K+ + [:S:]2−
(b) Ca and O
Ca → Ca2+ + 2e−
O + 2e− → O2−
Lewis representation:
Ca•• + :O:•••• → Ca2+ + [:O:]2−
(c) Al and N
Al → Al3+ + 3e−
N + 3e− → N3−
Lewis representation:
Al••• + :N:••• → Al3+ + [:N:]3−
👉 K → 1 valence electron
👉 Ca → 2 valence electrons
👉 Al → 3 valence electrons
👉 S → 6 valence electrons
👉 O → 6 valence electrons
👉 N → 5 valence electrons
Step 2: Electron transfer
👉 Metals lose electrons to form cations.
👉 Non-metals gain electrons to form anions.
Step 3: Formation of ionic compounds
• K2S is formed from K+ and S2−.
• CaO is formed from Ca2+ and O2−.
• AlN is formed from Al3+ and N3−.
👉 After gaining or losing electrons, atoms achieve a stable noble gas configuration.
👉 Lewis symbols are very useful for understanding ionic bond formation.
✅ In H2O, the O–H bond dipoles do not cancel because the molecule has a bent geometry. Hence, the molecule has a non-zero dipole moment.
👉 Dipole moment depends on the magnitude and direction of bond dipoles.
👉 If bond dipoles cancel each other, the molecule becomes non-polar.
(ii) Case of CO2
👉 CO2 has a linear geometry with a bond angle of 180°.
👉 The dipole moments of the two C=O bonds act in opposite directions.
👉 They cancel each other, giving net dipole moment = 0.
(iii) Case of H2O
👉 H2O has a bent or V-shaped geometry with a bond angle of about 104.5°.
👉 The O–H bond dipoles are not opposite to each other.
👉 Therefore, they do not cancel and a net dipole moment exists.
(iv) Conclusion
👉 CO2 → Linear and non-polar (μ = 0)
👉 H2O → Bent and polar (μ ≠ 0)
👉 Even if bonds are polar, the molecule can be non-polar if the dipoles cancel (as in CO2).
👉 Water is highly polar due to its bent structure and strong dipole moment.
Main applications:
✅ To determine whether a molecule is polar or non-polar.
✅ To estimate the percentage ionic character in a bond.
✅ To determine the molecular geometry or shape of molecules.
✅ To distinguish between molecules having similar structures but different polarity.
👉 If the dipole moment of a molecule is zero, the molecule is generally non-polar.
👉 If the dipole moment is non-zero, the molecule is polar.
(ii) Percentage ionic character
👉 Dipole moment helps in estimating the ionic character of a bond.
👉 A higher dipole moment generally indicates greater ionic character.
(iii) Determination of molecular geometry
👉 By measuring dipole moment, scientists can predict the arrangement of atoms in a molecule.
👉 Example:
• CO2 → μ = 0 → Linear structure.
• H2O → μ ≠ 0 → Bent structure.
(iv) Distinguishing isomers
👉 Dipole moment can differentiate between molecules that have the same formula but different structures.
👉 The formula for dipole moment is:
μ = q × d where q = magnitude of charge and d = distance between charges.
👉 Dipole moment helps chemists understand bond polarity and molecular structure.
✅ Electron gain enthalpy is the amount of energy released or absorbed when an isolated gaseous atom gains an electron to form a negative ion.
Main Difference:
👉 Electronegativity refers to atoms in a bonded state.
👉 Electron gain enthalpy refers to isolated gaseous atoms.
👉 It measures the ability of an atom to attract bonding electrons towards itself.
👉 It is a relative property and has no unit.
👉 Example: Fluorine has the highest electronegativity.
(ii) Electron Gain Enthalpy
👉 It is the energy change that occurs when an electron is added to an isolated gaseous atom.
👉 It is a measurable thermodynamic quantity.
👉 It is expressed in kJ mol−1.
(iii) Key differences
• Electronegativity → property of atoms in a molecule.
• Electron gain enthalpy → property of isolated gaseous atoms.
• Electronegativity → dimensionless value.
• Electron gain enthalpy → has energy units.
👉 Halogens usually have very high electron gain enthalpy because they need only one electron to complete their octet.
👉 Fluorine has the highest electronegativity value (≈ 4.0 on Pauling scale).
✅ As a result, one atom develops a partial negative charge (δ−) and the other develops a partial positive charge (δ+).
Example:
In HCl, chlorine is more electronegative than hydrogen, so the shared electron pair shifts towards chlorine.
Hδ+ — Clδ−
👉 In a covalent bond, atoms share electrons to achieve a stable electronic configuration.
(ii) Unequal sharing of electrons
👉 If the two atoms have different electronegativities, the shared electrons are pulled closer to the more electronegative atom.
👉 This unequal sharing results in partial charges on the atoms.
(iii) Example: HCl molecule
👉 Chlorine is more electronegative than hydrogen.
👉 Therefore, the bonding electrons shift towards chlorine.
👉 This creates a dipole in the molecule.
Hδ+ → Clδ−
👉 If the difference is very large, an ionic bond may form.
👉 Polar covalent bonds are common in molecules such as HCl, H2O, and NH3.
N2 < SO2 < ClF3 < LiF < K2O
👉 Difference in electronegativity (Δχ)
👉 Nature of atoms involved (metal or non-metal)
👉 Structure of the molecule
(i) N2
👉 Both atoms are identical, so
Δχ = 0
👉 Therefore it is a pure covalent bond and has the least ionic character.
(ii) SO2
👉 The electronegativity difference between S and O is moderate.
👉 Hence it is polar covalent and more ionic than N2.
(iii) ClF3
👉 The electronegativity difference between Cl and F is greater than in SO2.
👉 Therefore it has greater ionic character than SO2.
(iv) LiF
👉 Combination of metal (Li) and non-metal (F).
👉 The electronegativity difference is very large, so the compound is highly ionic.
(v) K2O
👉 Potassium is a very electropositive metal.
👉 When combined with oxygen, the ionic character becomes extremely high.
👉 Hence among the given compounds it shows the maximum ionic character.
Conclusion
Therefore, the increasing order of ionic character is:
N2 < SO2 < ClF3 < LiF < K2O
👉 Bonds between identical atoms such as N2 or O2 are pure covalent.
👉 Compounds formed between metals and non-metals usually show high ionic character.
H3C — C(=O) — O — H
👉 The second carbon atom forms a double bond with one oxygen (C=O) and a single bond with another oxygen.
👉 The second oxygen is bonded to hydrogen forming an –OH group.
👉 C = 4 electrons × 2 = 8
👉 O = 6 electrons × 2 = 12
👉 H = 1 electron × 4 = 4
👉 Total = 24 valence electrons.
Step 2: Skeleton structure
👉 Carbon atoms form the backbone of the molecule.
👉 The structure becomes:
CH3 – C – O – H
Step 3: Complete octets
👉 To satisfy the octet rule, one oxygen forms a double bond with carbon.
👉 The other oxygen forms single bonds with carbon and hydrogen.
Step 4: Lone pairs
👉 Each oxygen atom contains two lone pairs of electrons.
👉 This satisfies the octet rule for all atoms.
👉 The functional group present in acetic acid is the carboxyl group (–COOH).
👉 The C=O bond is shorter and stronger than the C–O bond.
✅ Carbon is a second period element and does not have d orbitals in its valence shell.
✅ Therefore, carbon cannot undergo dsp2 hybridisation.
✅ In CH4, carbon undergoes sp3 hybridisation, resulting in a tetrahedral geometry.
👉 In a square planar arrangement, four bonds lie in the same plane and the bond angles are 90°.
👉 This geometry generally arises from dsp2 hybridisation.
Step 2: Limitation of carbon
👉 Carbon has atomic number 6 and belongs to the second period.
👉 In the second period, only 2s and 2p orbitals are available.
👉 2d orbitals do not exist, so carbon cannot form dsp2 hybrid orbitals.
Step 3: Actual hybridisation in CH4
👉 In methane, carbon mixes one 2s orbital and three 2p orbitals to form sp3 hybrid orbitals.
👉 These four hybrid orbitals arrange themselves to minimize repulsion, forming a tetrahedral shape.
👉 The H–C–H bond angle is approximately 109.5°, not 90°.
Step 4: Conclusion
👉 Because carbon does not have d orbitals, it cannot adopt square planar geometry.
👉 Therefore, the stable geometry of CH4 is tetrahedral.
👉 The tetrahedral shape of CH4 is also explained by the VSEPR theory, where four bond pairs arrange themselves to minimize repulsion.
👉 Square planar geometry is commonly observed in some transition metal complexes, but not in methane.
✅ However, the BeH2 molecule has a linear geometry with a bond angle of 180°.
✅ Due to this symmetrical arrangement, the two bond dipole moments act in opposite directions and cancel each other.
👉 Therefore, the net dipole moment of BeH2 is zero.
👉 Hydrogen is slightly more electronegative than beryllium.
👉 Therefore, each Be–H bond has a bond dipole.
(ii) Molecular geometry
👉 According to VSEPR theory, BeH2 has two bonding pairs and no lone pairs around Be.
👉 The molecule therefore adopts a linear shape.
H — Be — H (180°)
(iii) Cancellation of dipole moments
👉 The dipole moments of the two Be–H bonds are equal and act in opposite directions.
👉 Hence they cancel each other.
(iv) Result
👉 Net dipole moment (μ) = 0.
👉 Similar examples include CO2 and CCl4.
👉 Dipole moment is therefore very useful in determining molecular geometry.
✅ In NH3, the dipole moments of the N–H bonds and the lone pair on nitrogen act in the same direction, increasing the overall dipole moment.
✅ In NF3, the dipole moments of the N–F bonds act in the opposite direction to the lone pair dipole, which reduces the net dipole moment.
👉 Both NH3 and NF3 have a trigonal pyramidal geometry due to the presence of one lone pair on nitrogen.
(ii) Case of NH3
👉 Nitrogen is more electronegative than hydrogen.
👉 Therefore, the bond dipoles of the N–H bonds point towards nitrogen.
👉 These dipoles act in the same direction as the lone pair dipole.
👉 This increases the net dipole moment.
(iii) Case of NF3
👉 Fluorine is more electronegative than nitrogen.
👉 The bond dipoles of N–F bonds point away from nitrogen.
👉 These dipoles act opposite to the lone pair dipole.
👉 Hence they partially cancel each other, resulting in a smaller dipole moment.
(iv) Result
NH3 → Dipole moment ≈ 1.46 D
NF3 → Dipole moment ≈ 0.24 D
👉 Dipole moment depends on both bond polarity and molecular geometry.
👉 Therefore, molecules with similar shapes can still have different dipole moments.
Types and shapes of hybrid orbitals:
• sp hybridisation → Linear shape (bond angle ≈ 180°)
• sp2 hybridisation → Trigonal planar shape (bond angle ≈ 120°)
• sp3 hybridisation → Tetrahedral shape (bond angle ≈ 109.5°)
👉 Atomic orbitals such as s and p orbitals combine to form new orbitals called hybrid orbitals.
👉 These hybrid orbitals have equal energy and identical shape.
(ii) sp hybridisation
👉 One s orbital mixes with one p orbital.
👉 Number of hybrid orbitals formed = 2.
👉 Arrangement → Linear geometry.
👉 Bond angle ≈ 180°.
Example: BeCl2, CO2.
(iii) sp2 hybridisation
👉 One s orbital mixes with two p orbitals.
👉 Number of hybrid orbitals formed = 3.
👉 Arrangement → Trigonal planar geometry.
👉 Bond angle ≈ 120°.
Example: BF3, SO3.
(iv) sp3 hybridisation
👉 One s orbital mixes with three p orbitals.
👉 Number of hybrid orbitals formed = 4.
👉 Arrangement → Tetrahedral geometry.
👉 Bond angle ≈ 109.5°.
Example: CH4.
👉 The number of hybrid orbitals formed is always equal to the number of orbitals mixed.
👉 Hybridisation is widely used to explain bonding in molecules like CH4, BF3, and BeCl2.
AlCl3 + Cl− → AlCl4−
• In AlCl3, hybridisation of Al = sp2.
• In AlCl4−, hybridisation of Al = sp3.
Therefore, the hybridisation changes as:
sp2 → sp3
👉 In AlCl3, aluminium forms three bonds with chlorine atoms.
👉 Therefore, there are three electron density regions around Al.
👉 This leads to sp2 hybridisation and a trigonal planar geometry.
Step 2: Addition of Cl−
👉 The chloride ion (Cl−) has a lone pair of electrons.
👉 It donates this electron pair to aluminium, forming a coordinate bond.
👉 Now aluminium becomes surrounded by four electron density regions.
Step 3: Hybridisation in AlCl4−
👉 With four bonding regions, aluminium undergoes sp3 hybridisation.
👉 The geometry becomes tetrahedral.
Step 4: Conclusion
👉 Thus, during the reaction the hybridisation of aluminium changes from
sp2 (in AlCl3) → sp3 (in AlCl4−) .
👉 The Cl− ion acts as a Lewis base by donating an electron pair.
👉 When hybridisation changes, the geometry of the molecule or ion also changes.
BF3 + NH3 → F3B·NH3
✅ When NH3 donates its lone pair to boron, a coordinate bond is formed.
👉 As a result, the hybridisation of boron changes from sp2 to sp3.
👉 The hybridisation of nitrogen remains sp3.
👉 Boron has three valence electrons and forms three σ-bonds with fluorine atoms.
👉 Therefore, boron undergoes sp2 hybridisation.
👉 Geometry → Trigonal planar.
(ii) Role of NH3
👉 Nitrogen in NH3 has a lone pair of electrons.
👉 This lone pair is donated to boron forming a coordinate (dative) bond.
(iii) Change in hybridisation
👉 After bond formation, boron becomes surrounded by four electron pairs.
👉 Therefore, boron changes its hybridisation to sp3 and the geometry becomes tetrahedral.
👉 Nitrogen already has sp3 hybridisation, so its hybridisation does not change.
👉 NH3 acts as a Lewis base because it donates a lone pair.
👉 The compound F3B·NH3 is called a Lewis acid–base adduct.
👉 Each carbon atom undergoes sp2 hybridisation.
👉 One σ (sigma) bond is formed by sp2–sp2 overlap between carbon atoms.
👉 One π (pi) bond is formed by sidewise overlap of unhybridised p-orbitals.
H H
\ /
C = C
/ \
H H
Double bond = 1 σ bond + 1 π bond
2. Formation of triple bond in C2H2 (Ethyne)
👉 Each carbon atom undergoes sp hybridisation.
👉 One σ bond is formed by sp–sp overlap between carbon atoms.
👉 Two π bonds are formed by overlap of two unhybridised p-orbitals.
H — C ≡ C — H
Triple bond = 1 σ bond + 2 π bonds
👉 Each carbon atom forms three σ bonds, so it undergoes sp2 hybridisation.
👉 The remaining unhybridised p-orbital forms a π bond.
Step 2: Hybridisation in C2H2
👉 Each carbon atom forms two σ bonds, so it undergoes sp hybridisation.
👉 Two unhybridised p-orbitals remain.
👉 These form two π bonds.
Step 3: Bond composition
👉 Double bond = 1 σ + 1 π.
👉 Triple bond = 1 σ + 2 π.
👉 π bonds are formed by sidewise overlap of p-orbitals.
👉 Triple bonds are shorter and stronger than double bonds.
(a) C2H2 (b) C2H4
Structure: H – C ≡ C – H
• Number of σ bonds = 3
• Number of π bonds = 2
(b) C2H4 (Ethene)
Structure: H2C = CH2
• Number of σ bonds = 5
• Number of π bonds = 1
👉 A sigma (σ) bond is formed by head-on overlap of orbitals.
👉 A pi (π) bond is formed by sidewise overlap of orbitals.
(ii) Bonds in C2H2
👉 One C≡C triple bond contains:
• 1 σ bond
• 2 π bonds
👉 Two C–H bonds contribute 2 σ bonds.
👉 Total = 3 σ and 2 π bonds.
(iii) Bonds in C2H4
👉 One C=C double bond contains:
• 1 σ bond
• 1 π bond
👉 Four C–H bonds contribute 4 σ bonds.
👉 Total = 5 σ and 1 π bond.
👉 A double bond contains 1 σ + 1 π bond.
👉 A triple bond contains 1 σ + 2 π bonds.
(a) 1s and 1s
(b) 1s and 2px
(c) 2py and 2py
(d) 1s and 2s
(c) 2py and 2py
because these orbitals are oriented perpendicular to the internuclear (x) axis and cannot overlap in a head-on manner.
The following combinations can form σ bonds:
• 1s – 1s
• 1s – 2px
• 1s – 2s
👉 A σ (sigma) bond forms when orbitals overlap head-on along the internuclear axis.
👉 In this question, the internuclear axis is the x-axis.
Step 2: Analysis of each pair
(a) 1s and 1s
👉 s orbitals are spherically symmetrical.
👉 They can overlap directly along the internuclear axis.
✅ Therefore, they form a σ bond.
(b) 1s and 2px
👉 The 2px orbital lies along the x-axis.
👉 Hence it can overlap head-on with the 1s orbital.
✅ Therefore, a σ bond is formed.
(c) 2py and 2py
👉 The 2py orbitals are oriented along the y-axis.
👉 Since the internuclear axis is x-axis, head-on overlap is not possible.
❌ Therefore, they cannot form a σ bond.
👉 Instead, they may overlap sidewise to form a π bond.
(d) 1s and 2s
👉 Both are s orbitals and are spherical.
👉 They can overlap along the internuclear axis.
✅ Therefore, a σ bond can form.
👉 If the axis is x-axis, then px forms σ overlap, while py and pz usually form π bonds.
👉 Because s orbitals are spherical, they can form σ bonds in almost any direction.
(a) CH3–CH3 (b) CH3–CH=CH2 (c) CH3–CH2–OH (d) CH3–CHO (e) CH3COOH
(b) In CH3–CH=CH2, the first carbon uses sp3 hybridisation while the second and third carbons use sp2 hybridisation.
(c) In CH3–CH2–OH, both carbon atoms have sp3 hybridisation.
(d) In CH3–CHO, the methyl carbon has sp3 hybridisation while the carbonyl carbon has sp2 hybridisation.
(e) In CH3COOH, the methyl carbon has sp3 hybridisation while the carboxyl carbon has sp2 hybridisation.
👉 If carbon forms four σ-bonds, hybridisation is sp3 and geometry becomes tetrahedral.
👉 If carbon forms three σ-bonds and one π-bond, hybridisation is sp2 with trigonal planar geometry.
Application to given molecules
(a) In ethane, each carbon forms four σ-bonds → sp3.
(b) In propene, the carbon atoms involved in C=C double bond are sp2 hybridised.
(c) In ethanol, all carbon atoms form only single bonds → sp3.
(d) In aldehyde group (–CHO), the carbon of C=O double bond is sp2 hybridised.
(e) In carboxylic acid group (–COOH), the carbonyl carbon is sp2 hybridised due to the C=O double bond.
👉 sp2 hybridised carbon forms trigonal planar geometry (120°).
👉 Double bonds always involve one σ-bond and one π-bond.
Example: In H2, the two hydrogen atoms share one pair of electrons. This shared pair is called a bond pair.
🔹 A lone pair is a pair of electrons present on one atom but not shared with another atom.
Example: In NH3, nitrogen has one lone pair of electrons which is not involved in bonding.
👉 When two atoms form a covalent bond, they share a pair of electrons.
👉 This shared pair is called a bond pair.
👉 Example: In H2, the bond between the two hydrogen atoms is formed by one shared electron pair.
(ii) Lone Pair
👉 Some electron pairs remain unshared on an atom.
👉 These non-bonding electrons are called lone pairs.
👉 Example: In NH3, nitrogen forms three bonds with hydrogen and keeps one lone pair.
👉 Lone pairs strongly influence molecular shape according to VSEPR theory.
👉 Due to this, the repulsion order in molecules is:
Lone pair – Lone pair > Lone pair – Bond pair > Bond pair – Bond pair .
👉 This is the reason why molecules like NH3 and H2O have distorted shapes.
✅ Generally, a σ bond is stronger, while a π bond is comparatively weaker.
| S.No. | Basis | σ Bond (Sigma Bond) | π Bond (Pi Bond) |
|---|---|---|---|
| 1 | Mode of formation | Formed by head-on overlap of orbitals. | Formed by sidewise overlap of orbitals. |
| 2 | Orbitals involved | Can be formed by s–s, s–p, p–p or hybrid orbitals. | Usually formed by unhybridised p–p orbitals (sometimes d orbitals). |
| 3 | Rotation | Free rotation around the bond axis is possible. | Free rotation is not possible because sidewise overlap would break. |
| 4 | Strength | Stronger and more stable. | Weaker and less stable. |
| 5 | Existence | Can exist independently (single bond = 1σ). | Cannot exist alone; it forms along with a σ bond. |
| 6 | Role in molecule | Determines the basic framework and geometry of the molecule. | Influences multiple bonding and reactivity. |
| 7 | Examples | Single bond = 1σ |
Double bond =
1σ + 1π Triple bond = 1σ + 2π |
👉 In a σ bond, electron density lies along the internuclear axis, making it stronger.
👉 π electrons are more reactive and therefore play an important role in many organic reactions.
✅ When the atoms approach each other, attractive and repulsive forces act simultaneously.
✅ At a certain distance, the net attraction becomes maximum and the energy becomes minimum, forming a stable H2 molecule.
✅ The bond length of H2 is about 74 pm and the bond dissociation energy is about 433 kJ mol−1.
👉 The Valence Bond Theory was proposed by Heitler and London (1927) and later developed by Pauling and others.
👉 This theory explains bond formation on the basis of orbital overlap and energy changes.
(ii) Initial state
Consider two hydrogen atoms:
👉 Atom A (nucleus NA, electron eA)
👉 Atom B (nucleus NB, electron eB)
When the atoms are far apart, there is no significant interaction between them.
(iii) Forces when atoms approach
Attractive forces:
NA–eA, NB–eB (internal attraction)
NA–eB, NB–eA (mutual attraction)
Repulsive forces:
eA–eB (electron–electron repulsion)
NA–NB (nucleus–nucleus repulsion)
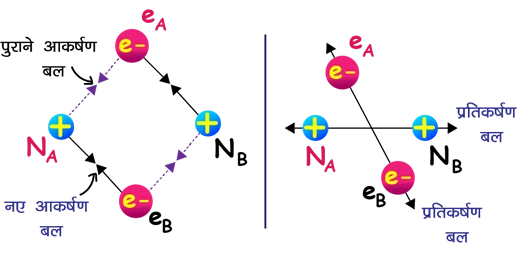
(iv) When does stability occur?
👉 As the atoms approach each other, the total energy decreases because attraction becomes stronger than repulsion.
👉 At a certain distance, attractive and repulsive forces balance each other.
👉 At this equilibrium distance the energy becomes minimum and a stable H2 molecule is formed.
👉 This distance is the bond length = 74 pm.
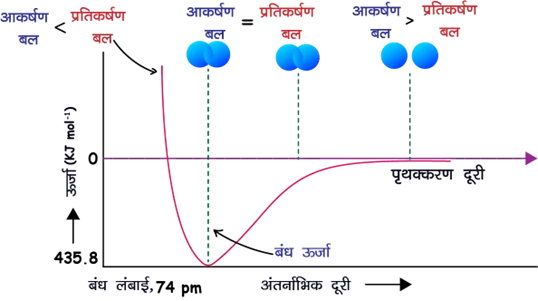
(v) Energy change
H(g) + H(g) → H2(g) + 433 kJ mol−1
👉 Energy is released during bond formation, making H2 more stable.
Reverse reaction:
H2(g) → H(g) + H(g) (ΔH = +433 kJ mol−1)
👉 This energy is called the bond dissociation energy.
(vi) Conclusion
✅ The H2 molecule is formed due to effective 1s–1s orbital overlap forming a σ bond.
✅ The minimum energy state corresponds to the maximum stability.
👉 The release of energy during H–H bond formation shows that bond formation is energetically favourable.
👉 The bond formed by 1s–1s axial overlap is called a σ bond.
✅ The energies of the combining atomic orbitals must be equal or nearly equal.
✅ The symmetry of the orbitals with respect to the molecular axis must be the same.
✅ There should be maximum overlap between the atomic orbitals.
👉 Atomic orbitals that combine to form molecular orbitals should have equal or nearly equal energies.
👉 If the energy difference between orbitals is very large, effective combination does not occur.
Example:
👉 In the same atom, 1s–1s overlap is possible.
👉 The energy difference between 1s and 2s is very large, so effective combination is unlikely.
👉 In different atoms, the orbitals should have approximately similar energies.
2) Similar Symmetry
👉 The combining orbitals must have the same symmetry with respect to the molecular axis (usually the z-axis).
👉 If symmetry does not match, overlap cannot occur, even if the energies are similar.
Example:
👉 2pz can combine with 2pz.
👉 2pz cannot combine with 2px or 2py because their symmetry is different.
3) Maximum Overlap
👉 The greater the overlap between atomic orbitals, the stronger the interaction.
👉 Greater overlap increases the electron density between nuclei, leading to a more stable bonding molecular orbital.
Conclusion
For effective LCAO formation, all three conditions must be satisfied:
Similar Energy + Same Symmetry + Maximum Overlap
Only then a stable bonding molecular orbital is formed.
👉 Greater orbital overlap usually means a stronger chemical bond.
👉 In Molecular Orbital Theory, properties such as bond order and magnetic behaviour are explained using these LCAO conditions.
Since the bond order is zero, there is no net bond formation between the two beryllium atoms.
Therefore, the Be2 molecule does not exist as a stable molecule.
👉 Beryllium has atomic number 4.
👉 Electronic configuration: 1s2 2s2.
(ii) Molecular orbital formation
👉 When two Be atoms combine, their 2s atomic orbitals overlap to form:
• σ2s (bonding orbital)
• σ*2s (antibonding orbital)
(iii) Electron filling
👉 Total valence electrons in Be2 = 4 electrons.
👉 Distribution of electrons:
σ2s → 2 electrons
σ*2s → 2 electrons
(iv) Bond order calculation
👉 Bond order = ½ (Number of bonding electrons − Number of antibonding electrons)
👉 Bond order = ½ (2 − 2) = 0.
👉 Since the bond order is zero, no stable bond is formed.
👉 If bond order = 0, the molecule is unstable and does not exist.
👉 Molecules like H2, O2, and N2 exist because their bond order is greater than zero.
O2, O2+, O2- (superoxide), O22− (peroxide)
Bond order values:
O2+ → 2.5
O2 → 2
O2- → 1.5
O22− → 1
Relative stability:
O2+ > O2 > O2- > O22−
Magnetic properties:
O2 → Paramagnetic
O2+ → Paramagnetic
O2- → Paramagnetic
O22− → Diamagnetic
👉 Oxygen molecule has 16 electrons.
👉 In MO theory, the last two electrons enter the π* antibonding orbitals.
Step 2: Bond order formula
👉 Bond Order = (Bonding electrons − Antibonding electrons) / 2
Step 3: Bond order of O2
👉 Bond order = 2.
👉 Two unpaired electrons → Paramagnetic.
Step 4: O2+
👉 One electron removed from antibonding orbital.
👉 Antibonding electrons decrease.
👉 Bond order becomes 2.5.
👉 One unpaired electron → Paramagnetic.
Step 5: O2- (Superoxide)
👉 One electron added to antibonding orbital.
👉 Bond order decreases to 1.5.
👉 One unpaired electron → Paramagnetic.
Step 6: O22− (Peroxide)
👉 Two electrons added to antibonding orbitals.
👉 Bond order becomes 1.
👉 All electrons paired → Diamagnetic.
Step 7: Stability comparison
👉 Higher bond order means stronger bond and greater stability.
👉 Therefore stability order is:
O2+ > O2 > O2- > O22−
👉 According to simple bonding theories, oxygen should be diamagnetic, but experiments show it is paramagnetic.
👉 This difference was explained correctly only by MO theory.
✅ These signs indicate the phase (sign) of the wave function of the orbital.
✅ When orbitals with the same phase overlap, a bonding molecular orbital is formed.
✅ When orbitals with opposite phases overlap, an antibonding molecular orbital is formed.
👉 The + and – signs on the lobes of atomic orbitals are mathematical symbols representing the phase of the wave function.
👉 They do not indicate positive or negative electric charge.
(ii) Same sign overlap (in-phase overlap)
👉 When two orbitals overlap with the same phase (+ with + or – with –), constructive overlap occurs.
👉 Electron density between the nuclei increases.
✅ Result: Formation of a bonding molecular orbital.
(iii) Opposite sign overlap (out-of-phase overlap)
👉 When orbitals overlap with opposite phases (+ with –), destructive overlap occurs.
👉 Electron density between nuclei decreases and a node may form.
✅ Result: Formation of an antibonding molecular orbital.
(iv) Conclusion
👉 The + and – signs determine whether orbital overlap will produce a bonding or antibonding molecular orbital.
Same phase → Bonding MO
Opposite phase → Antibonding MO
👉 Changing the phase relationship between orbitals can completely change the result of orbital overlap.
👉 Phase relationships are very important in understanding the nature and strength of chemical bonds.
This hybridisation produces five hybrid orbitals arranged in a trigonal bipyramidal geometry.
👉 Three bonds lie in the same plane and are called equatorial bonds.
👉 Two bonds lie perpendicular to this plane and are called axial bonds.
The axial bonds are longer and weaker than equatorial bonds because they experience greater repulsion.
👉 Atomic number of phosphorus = 15.
👉 Valence shell configuration = 3s2 3p3.
👉 One electron from the 3s orbital is promoted to the 3d orbital.
👉 Now phosphorus has five unpaired electrons which form five P–Cl σ-bonds.
(ii) Geometry of PCl5
👉 The molecule has trigonal bipyramidal geometry.
👉 Bond angles:
Equatorial–Equatorial = 120°
Axial–Equatorial = 90°
Axial–Axial = 180°
(iii) Reason for longer axial bonds
👉 Each axial bond experiences repulsion from three equatorial bond pairs.
👉 Due to this greater bond pair–bond pair repulsion, axial bonds become longer and weaker than equatorial bonds.
👉 That is why larger atoms or lone pairs generally occupy equatorial positions.
👉 The molecule PCl5 exists as a gas or liquid, but in solid state it forms [PCl4]+ and [PCl6]− ions.
✅ A hydrogen bond is generally stronger than van der Waals forces but weaker than a covalent bond.
👉 When hydrogen is covalently bonded to a highly electronegative atom such as F, O, or N, the hydrogen atom acquires a partial positive charge (δ+).
👉 This partially positive hydrogen is attracted to a lone pair of electrons on another electronegative atom.
👉 This special intermolecular attraction is called a hydrogen bond.
(ii) Examples
👉 In water: O–H···O
👉 In hydrogen fluoride: H–F···F
👉 In ammonia: N–H···N
(iii) Comparison with van der Waals forces
👉 Hydrogen bonding is stronger than ordinary van der Waals forces because it involves stronger dipole–dipole attraction.
👉 However, it is still weaker than a covalent bond.
👉 The open crystal structure of ice is also due to hydrogen bonding.
👉 Hydrogen bonding plays an important role in stabilizing biological molecules such as DNA and proteins.
N2, O2, O2+, O2−
Formula:
Bond Order = (Bonding electrons − Antibonding electrons) / 2
Bond order values:
N2 → 3
O2 → 2
O2+ → 2.5
O2− → 1.5
👉 Bond order is calculated using the formula:
Bond Order = (Nb − Na) / 2
where:
Nb = number of bonding electrons
Na = number of antibonding electrons
Step 2: Bond order of N2
👉 Total electrons = 14.
👉 Bonding electrons = 10.
👉 Antibonding electrons = 4.
👉 Bond order = (10 − 4) / 2 = 3.
Step 3: Bond order of O2
👉 Total electrons = 16.
👉 Bonding electrons = 10.
👉 Antibonding electrons = 6.
👉 Bond order = (10 − 6) / 2 = 2.
Step 4: Bond order of O2+
👉 One electron is removed from the antibonding orbital.
👉 Antibonding electrons become 5.
👉 Bond order = (10 − 5) / 2 = 2.5.
Step 5: Bond order of O2−
👉 One electron is added to an antibonding orbital.
👉 Antibonding electrons become 7.
👉 Bond order = (10 − 7) / 2 = 1.5.
👉 For example, N2 has bond order 3, so it has a very strong triple bond.
👉 Species with higher bond order are more stable.
