
Class 12 Chemistry Chapter -2 Electrochemistry Notes [English Medium]Updated – MyNoteswala
Highlight of the chapter.
- Describe an electrochemical cell and differentiate between galvanic and electrolytic cells.
- Standard H- electrode (SHE).
- Electrochemical series.
- Apply Nernst equation for calculating the emf of galvanic cell and define standard potential of the cell.
- Derive relation between standard potential of the cell, Gibbs energy of cell reaction and its equilibrium
constant. - Define resistivity , conductivity and molar conductivity (λm) of ionic solutions.
- Differentiate between ionic (electrolytic) and electronic conductivity;
- Measurement of conductivity of electrolytic solutions and calculation of their molar conductivity;
- Explanation of variation of conductivity and molar conductivity of solutions with change in their concentration and define molar conductivity at zero concentration or infinite dilution).
- Kohlrausch law and its applications.
- Describe the construction of some primary and secondary batteries and fuel cells.
- Explain corrosion as an electrochemical process (Rusting of iron).


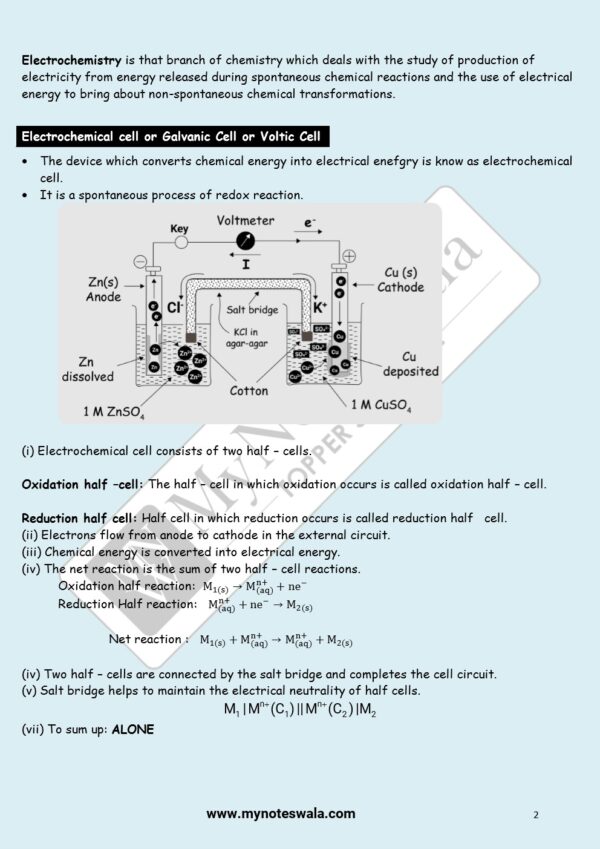
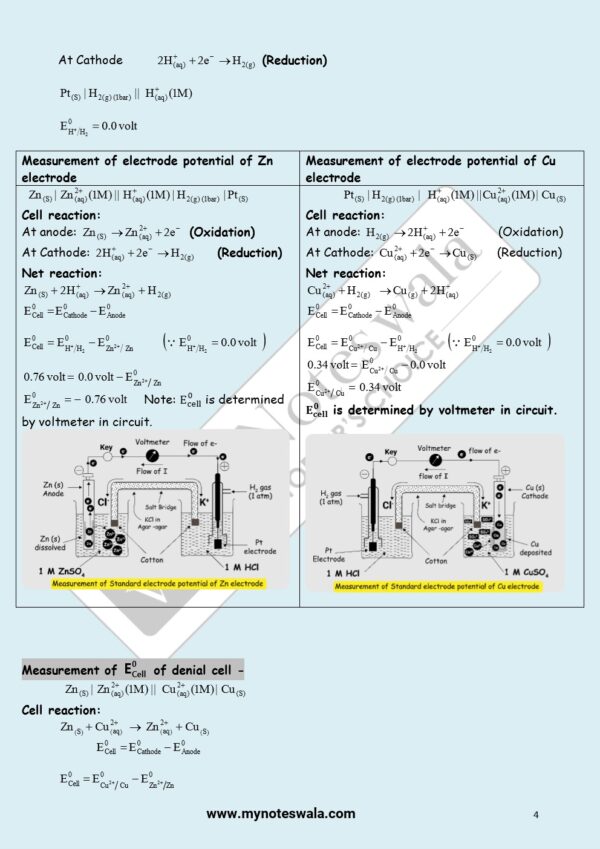












![Class 12 Chemistry Chapter 4 d- and f- Block Elements [English Medium] – MyNoteswala](https://mynoteswala.com/wp-content/uploads/2023/07/Class-12-Chemistry-chapter-4-English-Medium-300x300.webp)
![Class 12 Chemistry Chapter 8 Aldehyde, Ketone and Carboxylic Acids [English Medium] PDF – MyNoteswala](https://mynoteswala.com/wp-content/uploads/2024/01/Unit-8-300x300.png)
![Class 12 Chemistry Chapter 1 to 10 [English Medium] – MyNoteswala](https://mynoteswala.com/wp-content/uploads/2023/08/Class-12-Chemistry-all-chapters-English-medium-300x300.webp)
![Class 12 Chemistry Chapter 1 Solutions [English Medium] PDF – MyNoteswala](https://mynoteswala.com/wp-content/uploads/2024/02/Unit-1-Solution-300x300.png)
![Class 12 Chemistry Chapter 1 to 10 [Hindi Medium] PDF – MyNoteswala](https://mynoteswala.com/wp-content/uploads/2024/05/Class-12-Chemistry-All-units-Hindi-pdf-300x300.png)
![Class 12 Chemistry Chapter 10 Biomolecules Notes [English Medium] pdf– MyNoteswala](https://mynoteswala.com/wp-content/uploads/2023/10/Unit-10-300x300.png)
![Class 12 Chemistry Chapter 2 Electrochemistry [English Medium] PDF – MyNoteswala](https://mynoteswala.com/wp-content/uploads/2024/02/Unit-2-Electrochemistry-300x300.png)
Mukesh Rajpurohit (verified owner) –
Good quality.
Suraj Jaiswal (verified owner) –
Good quality.
Vivek Verma (verified owner) –
Good quality.
Sneha Saxena (verified owner) –
Good quality.
Ritu singh (verified owner) –
Good quality.
Pramod Soni (verified owner) –
Good quality.
Rajini Rathore (verified owner) –
Good quality.
Surbhi Sati (verified owner) –
Good quality.
Lokesh Khadiya (verified owner) –
Good quality.
Sarita Chaube (verified owner) –
Good quality.
Manoj Kesharwani (verified owner) –
Good quality.
Golu Singh (verified owner) –
Good quality.
Kanupriya Sharma (verified owner) –
Good quality.
Radhika Sherawat (verified owner) –
Good quality.
Lokesh Saini (verified owner) –
Good quality.
Munna Yadav (verified owner) –
Good quality.
Neelam Pandey (verified owner) –
Good quality.
Suman pal (verified owner) –
Good quality.
Abhishek Sharma (verified owner) –
Good quality.
Gudiya Gupta (verified owner) –
Good quality.
Kunal Singh (verified owner) –
Good quality.
Rupali Roy (verified owner) –
Good quality.
Kritika Singh (verified owner) –
Good quality.
Nihit Naithani (verified owner) –
Good quality.
Sajiya Khan (verified owner) –
Good quality.
Subhash Shahu (verified owner) –
Good quality.
Sadiq ali (verified owner) –
Good quality.
Pukhraj Meena (verified owner) –
Good quality.
Sabita Kumari (verified owner) –
Good quality.
Neha Savita (verified owner) –
Good quality.
Harshit Kumar (verified owner) –
Good quality.
Abhay Sharma (verified owner) –
Good quality.
Sabita Sharma (verified owner) –
Good quality.
Rita Saxena (verified owner) –
Good quality.
Sandhya Sharma (verified owner) –
Good quality.
Mukesh Bansal (verified owner) –
Good quality.
Akash Sharma (verified owner) –
Good quality.
Vimu Saini (verified owner) –
Good quality.
Kusum Gupta (verified owner) –
Good quality.
Kunal Singh (verified owner) –
Good quality.
Monty singh (verified owner) –
Good quality.
Lakshmi Rawat (verified owner) –
Good quality.
Jignash Patel (verified owner) –
Good quality.
Kusum Gupta (verified owner) –
Good quality.
Riya kumari (verified owner) –
Good quality.
Aman Bhardwaj (verified owner) –
Good quality.
Kritika Singh (verified owner) –
Good quality.
Shiva Singh (verified owner) –
Good quality.
Sarita Chaube (verified owner) –
Good quality.
Sonali Kothwal (verified owner) –
Good quality.
Lokesh Gupta (verified owner) –
Good quality.
Saloni Singh (verified owner) –
Good quality.
Himanshu Upadhyay (verified owner) –
Good quality.
Pramod Sharma (verified owner) –
Good quality.
Ritu singh (verified owner) –
Good quality.
Narayni Sharma (verified owner) –
Good quality.
Neha Savita (verified owner) –
Good quality.
Siddharth pandit (verified owner) –
Good quality.
Prachi Sahgal (verified owner) –
Good quality.
Anshra Kumari (verified owner) –
Good quality.
Sandhya Verma (verified owner) –
Good quality.
Munna Yadav (verified owner) –
Good quality.
Nitya Singh (verified owner) –
Good quality.
Tejinder singh (verified owner) –
Good quality.
Suraj Jaiswal (verified owner) –
Good quality.
Rajendra Sharma (verified owner) –
Good quality.
Usha Tripathi (verified owner) –
Good quality.
Lokesh Gupta (verified owner) –
Good quality.
Anuj Sherawat (verified owner) –
Good quality.
Gungun Sharma (verified owner) –
Good quality.
Pukhraj Meena (verified owner) –
Good quality.
Madhulika Hirwar (verified owner) –
Good quality.
Dinesh Sharma (verified owner) –
Good quality.
Sandhya Verma (verified owner) –
Good quality.
Ruby Singh (verified owner) –
Good quality.
Kaushal Singh (verified owner) –
Good quality.
Khushi Saxena (verified owner) –
Good quality.
Yogesh Dakar (verified owner) –
Good quality.
Yogesh Dakar (verified owner) –
Good quality.
Manoj Goel (verified owner) –
Good quality.
Mayank Kumar (verified owner) –
Good quality.
Chetana Sharma (verified owner) –
Good quality.
Santosh Singh (verified owner) –
Good quality.
Manish Kumar (verified owner) –
Good quality.
Swati Jain (verified owner) –
Good quality.
Udai Singh (verified owner) –
Good quality.
Prachi Sahgal (verified owner) –
Good quality.
Manish Kumar (verified owner) –
Good quality.
Ankur pandit (verified owner) –
Good quality.
Shivam Chaudhary (verified owner) –
Good quality.
Kusum Gupta (verified owner) –
Good quality.
Madhulika Hirwar (verified owner) –
Good quality.
Manish Kumar (verified owner) –
Good quality.
Vipin Singh (verified owner) –
Good quality.
Rita Saxena (verified owner) –
Good quality.
Babita Ghosh (verified owner) –
Good quality.
Aditi Sharma (verified owner) –
Good quality.
Pramod Soni (verified owner) –
Good quality.
Swati Sinha (verified owner) –
Good quality.
Pramod Sharma (verified owner) –
Good quality.
Niharika Jain (verified owner) –
Good quality.
Pramod Soni (verified owner) –
Good quality.
Prachi Kumari (verified owner) –
Good quality.
Kundan Kumar (verified owner) –
Good quality.
Kundan Kumar (verified owner) –
Good quality.
Anupam Sharma (verified owner) –
Good quality.
Chetana Sharma (verified owner) –
Good quality.
Aashu kumar (verified owner) –
Good quality.
Kunal Maurya (verified owner) –
Good quality.
Rajini Rathore (verified owner) –
Good quality.
Raunak Sharma (verified owner) –
Good quality.
Gudiya Gupta (verified owner) –
Good quality.
Swati Jain (verified owner) –
Good quality.
Lokesh Khadiya (verified owner) –
Good quality.
Mukesh Kumar (verified owner) –
Good quality.
Anshra Kumari (verified owner) –
Good quality.
Depali Sharma (verified owner) –
Good quality.
Ritu singh (verified owner) –
Good quality.
Anita rao (verified owner) –
Good quality.
Aditi Agarwal (verified owner) –
Good quality.
Lokesh Khadiya (verified owner) –
Good quality.
Ajit Biswal (verified owner) –
Good quality.
Santosh Singh (verified owner) –
Good quality.
Usha Tripathi (verified owner) –
Good quality.
Harsh Kumawat (verified owner) –
Good quality.
Vedant patil (verified owner) –
Good quality.
Anupam Sharma (verified owner) –
Good quality.
Dinesh Sharma (verified owner) –
Good quality.
Pramod Soni (verified owner) –
Good quality.
Mayank Gupta (verified owner) –
Good quality.
Lakshmi Rawat (verified owner) –
Good quality.
Chetana Saini (verified owner) –
Good quality.
Tejinder singh (verified owner) –
Good quality.
Pankaj Gupta (verified owner) –
Good quality.
Mayank Patel (verified owner) –
Good quality.
Manoj Kesharwani (verified owner) –
Good quality.
Manisha Sharma (verified owner) –
Good quality.
Tejinder singh (verified owner) –
Good quality.
Saloni Singh (verified owner) –
Good quality.
Shivam Chaudhary (verified owner) –
Good quality.
Chetana Sharma (verified owner) –
Good quality.
Sandhya Singh (verified owner) –
Good quality.
Kavita Sharma (verified owner) –
Good quality.
Santosh Singh (verified owner) –
Good quality.
Amisha Gupta (verified owner) –
Good quality.
Swati Sinha (verified owner) –
Good quality.
Sk Dhamani (verified owner) –
Good quality.
Swati Jain (verified owner) –
Good quality.
Vaidehi (verified owner) –
Good quality.
Swati Sinha (verified owner) –
Good quality.
Ajit Biswal (verified owner) –
Good quality.
Karnika Goswami (verified owner) –
Good quality.
Mayank Choudhary (verified owner) –
Good quality.
Pukhraj Meena (verified owner) –
Good quality.
Pawan Agarwal (verified owner) –
Good quality.
Narayni Sharma (verified owner) –
Good quality.
Manoj Goel (verified owner) –
Good quality.
Sneha Saxena (verified owner) –
Good quality.
Anoushka gupta (verified owner) –
Thankyou mynoteswala…. it helped me a lot.
Gungun Sharma (verified owner) –
Good quality.
Monu Yadav (verified owner) –
Good quality.
Himanshu Upadhyay (verified owner) –
Good quality.
Mohit Saxena (verified owner) –
Good quality Notes
Lokesh Sharma (verified owner) –
Good quality.
Anita rao (verified owner) –
Good quality.
Chetan Patil (verified owner) –
Good quality.
Mayank Choudhary (verified owner) –
Good quality.
Lokesh Khadiya (verified owner) –
Good quality.
Rajendra Joshi (verified owner) –
Good quality.
Aarti Kumari (verified owner) –
Chemistry made simple… go for it.
Anchal kumari (verified owner) –
Wonderful 👍
Mohit Sing (verified owner) –
Nice Notes…. Very Helpful
Pramod Mishra (verified owner) –
Good quality.
Anupam Sharma (verified owner) –
Good quality.
Harshita Meena (verified owner) –
Low price …. High return
Pakaj Jain (verified owner) –
Notes + Youtube video ka tadka … Maja aagya
Kavita Sharma (verified owner) –
Good quality.
Kusum Gupta (verified owner) –
Good quality.
Shrikant (verified owner) –
GREAT Notes
Lokesh Saini (verified owner) –
Good quality.
Shivam kumar Gupta (verified owner) –
Good quality.
Indu kumari (verified owner) –
GREAT Notes
Payel Joshi (verified owner) –
Good quality.
Tashi Gurjar (verified owner) –
Super se bhi upar wale notes….☺☺☺
Kritika Saini (verified owner) –
Good quality.
Vipin Singh (verified owner) –
Good quality.
Saloni Singh (verified owner) –
Good quality.
Komal Soni (verified owner) –
Thankyou mynoteswala…. it helped me a lot.
Gungun Sharma (verified owner) –
Good quality.
Nihit Naithani (verified owner) –
Good quality.
Pankaj Gupta (verified owner) –
Very Helpful
Komal Soni (verified owner) –
Nice Notes…. Very Helpful
Shrikant (verified owner) –
Awesome Notes
Pakaj Jain (verified owner) –
Simple explained
Sabita Kumari (verified owner) –
Good quality.
Anoushka gupta (verified owner) –
Thankyou mynoteswala…. it helped me a lot.
Mayank Kumar (verified owner) –
Good quality.
Rupendra Yadav (verified owner) –
Good quality.
Siddharth pandit (verified owner) –
Good quality.
Lokesh Beniwal (verified owner) –
Great notes… buy again
Vaidehi (verified owner) –
Good quality.
Payel Joshi (verified owner) –
Good quality.
Mohit Gujar (verified owner) –
Awesome Notes
Kuldeep Singh (verified owner) –
Simple notes but powerful
Komal Soni (verified owner) –
Excellent for students
Aditi Sharma (verified owner) –
Good quality.
Vijay Malawat (verified owner) –
Nice notes + tricks
Pooja Sharma (verified owner) –
Good quality.
Vishal Jangid (verified owner) –
Good quality.
Pankaj Gupta (verified owner) –
Wonderful 👍
Harshita Meena (verified owner) –
Nice Notes…. Very Helpful
Neha Yadav (verified owner) –
Best Notes…
Shiva Singh (verified owner) –
Good quality.
Rupali Roy (verified owner) –
Good quality.
Parul Sharma (verified owner) –
Awesome notes
Sk Dhamani (verified owner) –
Good quality.
Nikita Agarwal (verified owner) –
Nice Notes…. Very Helpful
Harish Rathore (verified owner) –
Good quality.
Siddharth pandit (verified owner) –
Good quality.
Ritu singh (verified owner) –
GREAT Notes
Ayen patel (verified owner) –
Good quality.
Usha Saini (verified owner) –
Good quality.
Manish Kumar (verified owner) –
Good quality.
Harshita Meena (verified owner) –
GREAT Notes
Mukesh Rajpurohit (verified owner) –
Good quality.
Lakshmi Rawat (verified owner) –
Good quality.
Usha Saini (verified owner) –
Good quality.
Diya Yadav (verified owner) –
Good quality.
Suresh Kumar (verified owner) –
The Best
Rajendra Joshi (verified owner) –
Good quality.
Subhash Shahu (verified owner) –
Good quality.
Niharika Jain (verified owner) –
Notes + Youtube video ka tadka … Maja aagya
Pukhraj Meena (verified owner) –
Good quality.
Babita Ghosh (verified owner) –
Good quality.
Aarti Kumari (verified owner) –
Excellent for students
Indu kumari (verified owner) –
I found it helpful 👍
Raunak Sharma (verified owner) –
Good quality.
Pramod Soni (verified owner) –
Good quality.
Sandhya Sharma (verified owner) –
Good quality.
Siddharth pandit (verified owner) –
Good quality.
Pakaj Jain (verified owner) –
Simple explained
Nishant Yadav (verified owner) –
Good quality.
Lokesh Saini (verified owner) –
Good quality.
Mukesh Rajpurohit (verified owner) –
Good quality.
Manish Vaswani (verified owner) –
Simple notes but powerful
Sarita Chaube (verified owner) –
Good quality.
Shiva Singh (verified owner) –
Good quality.
Santosh Singh (verified owner) –
Good quality.
Harshita Meena (verified owner) –
Good for revision .. ☺
Santosh Singh (verified owner) –
Good quality.
Nikhil Yadav (verified owner) –
Very affordable
Nitya Singh (verified owner) –
Good quality.
Arushi Kumari (verified owner) –
Good quality.
Akash Sharma (verified owner) –
Good quality.
Sadiq ali (verified owner) –
Good quality.
Ajit Biswal (verified owner) –
Good quality.
Vishal Yadav (verified owner) –
Good quality Notes
Vijay Malawat (verified owner) –
GREAT Notes
Vishal Jangid (verified owner) –
Good quality.
Sonu Saini (verified owner) –
Good quality.
Pankaj Gupta (verified owner) –
Only 29/- wow
Sabita Kumari (verified owner) –
Good quality.
Shrikant (verified owner) –
Concept was well explained.
Diya Yadav (verified owner) –
Good quality.
Sanjana Shrimal (verified owner) –
Very affordable
Harshita Meena (verified owner) –
Loved it…. Buy again
Manish Vaswani (verified owner) –
Best Notes…
Niharika Jain (verified owner) –
Good quality.
Suresh Kumar (verified owner) –
Perfect notes
Vipin Singh (verified owner) –
Good quality.
Lokesh Gupta (verified owner) –
Good quality.
Raunak Sharma (verified owner) –
Good quality.
Arushi Kumari (verified owner) –
Good quality.
Himanshu Joshi (verified owner) –
Good quality.
Abhishek Sharma (verified owner) –
Good quality.
Payel Joshi (verified owner) –
Concept clearing notes
Kunal Maurya (verified owner) –
Good quality.
Amit raj (verified owner) –
Good quality.
Kundan Jha (verified owner) –
Good quality.
Sarita Sharma (verified owner) –
Good quality.
Yogesh Meena (verified owner) –
It helps
Lokesh Khadiya (verified owner) –
Good quality.
Rahul Sharma (verified owner) –
Easily explained
Anoushka gupta (verified owner) –
It helps
Sadiq ali (verified owner) –
Good quality.
Shubham kurade (verified owner) –
Good quality.
Payel Joshi (verified owner) –
Good quality.
Niharika Jain (verified owner) –
Told my friend about these notes
Rupendra Yadav (verified owner) –
Good quality.
Nikita Sharma (verified owner) –
Bahut Acche notes hai
Purva Soni (verified owner) –
Good quality.
Nikhil Yadav (verified owner) –
Perfect hai bhai
Rajendra Joshi (verified owner) –
Good quality.
Vijay Daswani (verified owner) –
GREAT Notes
Mihir patel (verified owner) –
Good quality.
Kundan Jha (verified owner) –
Good quality.
Gudiya Gupta (verified owner) –
Good quality.
Anuj Sherawat (verified owner) –
Good quality.
Shivam Chaudhary (verified owner) –
Good quality.
Niharika Jain (verified owner) –
Good quality.
Ayen patel (verified owner) –
Good quality.
Manish Vaswani (verified owner) –
Concept clearing notes
Manoj Kesharwani (verified owner) –
Good quality.
Kavita Sharma (verified owner) –
Good quality.
Gungun Sharma (verified owner) –
Good quality.
Mohit Sing (verified owner) –
Loved it…. Buy again
Pawan Agarwal (verified owner) –
Nice Notes…. Very Helpful
Nikita Sharma (verified owner) –
Very Helpful
Kanupriya Sharma (verified owner) –
Good quality.
Sanjana Shrimal (verified owner) –
It helps
Arushi Kumari (verified owner) –
Good quality.
Sk Dhamani (verified owner) –
Good quality.
Rajendra Joshi (verified owner) –
Good quality.
Golu Singh (verified owner) –
Good quality.
Chetana Sharma (verified owner) –
Good quality.
Sanjana Shrimal (verified owner) –
Easily explained
Vijay Rathore (verified owner) –
Good quality.
Sandhya Yadav (verified owner) –
Good quality.
Ravi Sharma (verified owner) –
Low price …. High return
Sandhya Verma (verified owner) –
Good quality.
Nikita Sharma (verified owner) –
Recommended
Rakhi Saini (verified owner) –
Chemistry made simple… go for it.
Ritu singh (verified owner) –
The Best
Monty singh (verified owner) –
Good quality.
Pramod Sharma (verified owner) –
Good quality.
Pramod Sharma (verified owner) –
Good quality.
Subhash Shahu (verified owner) –
Good quality.
Sanjana Shrimal (verified owner) –
GREAT Notes
Lokesh Saini (verified owner) –
Good quality.
Suman pal (verified owner) –
Good quality.
Vijay Daswani (verified owner) –
Awesome notes
Saloni Singh (verified owner) –
Good quality.
Vivek Verma (verified owner) –
Good quality.
Anuj Sherawat (verified owner) –
Good quality.
Manish Vaswani (verified owner) –
Very Helpful
Abhay Sharma (verified owner) –
Good quality.
Mohit Sing (verified owner) –
Nice Notes…. Very Helpful
Shivam Chaudhary (verified owner) –
Good quality.
Shivam Chaudhary (verified owner) –
Good quality.
Rajiv Bhattacharjee (verified owner) –
Good quality.
Shivam Chaudhary (verified owner) –
Good quality.
Sonali Kothwal (verified owner) –
Good quality.
Mohit Gujar (verified owner) –
Simple notes but powerful
Vijay Daswani (verified owner) –
Good one… 👍
Shivam kumar Gupta (verified owner) –
Good quality.
Kavita Sharma (verified owner) –
Very affordable
Aarti Kumari (verified owner) –
Wonderful 👍
Udai Singh (verified owner) –
Good quality.
Rahul Sharma (verified owner) –
Wonderful 👍
Trisha Gupta (verified owner) –
Nice notes + tricks
Kavita Soni (verified owner) –
Loved it
Neelam Pandey (verified owner) –
Good quality.
Shrikant (verified owner) –
Good for revision .. ☺
Priya Joshi (verified owner) –
I found it helpful 👍
Nihit Naithani (verified owner) –
Good quality.
Kuldeep Kumar (verified owner) –
Good quality.
Rita Saxena (verified owner) –
Good quality.
Rakhi Saini (verified owner) –
Perfect notes
Monty singh (verified owner) –
Nice one
Harsh Kumawat (verified owner) –
Good quality.
Rohit kumar (verified owner) –
The Best
Pankaj Goyal (verified owner) –
Nice notes + tricks
Pakaj Jain (verified owner) –
Very Helpful
Payel Joshi (verified owner) –
Low price …. High return
Kritika Singh (verified owner) –
Good quality.
Rakhi Saini (verified owner) –
Good one… 👍
Pakaj Jain (verified owner) –
Bahut Acche notes hai
Anupam Sharma (verified owner) –
Good quality.
Manoj Kesharwani (verified owner) –
Good quality.
Indresh Kumar (verified owner) –
Good quality.
Pramod Soni (verified owner) –
Good quality.
Indu kumari (verified owner) –
Loved it…. Buy again
Vijay Daswani (verified owner) –
Loved it…. Buy again
Anoushka gupta (verified owner) –
Super se bhi upar wale notes….☺☺☺
Rohit kumar (verified owner) –
Excellent for students
Amisha Gupta (verified owner) –
Good quality.
Vishal Yadav (verified owner) –
Good one… 👍
Mohit Gujar (verified owner) –
Excellent for students
Monty singh (verified owner) –
Very affordable
Madhulika Hirwar (verified owner) –
Good quality.
Pankaj Goyal (verified owner) –
Only 29/- wow
Pawan Agarwal (verified owner) –
Good quality.
Abhay Sharma (verified owner) –
Good quality.
Suresh Kumar (verified owner) –
Simple notes but powerful
Rajendra Joshi (verified owner) –
Good quality.
Lokesh Khadiya (verified owner) –
Good quality.
Pakaj Jain (verified owner) –
GREAT Notes
Harshita Meena (verified owner) –
Notes + Youtube video ka tadka … Maja aagya
Lokesh Saini (verified owner) –
Good quality.
Indu kumari (verified owner) –
Bahut Acche notes hai
Vijay Agarwal (verified owner) –
It helps
Radhika Sherawat (verified owner) –
Good quality.
Dinesh Sharma (verified owner) –
Good quality.
Payel Joshi (verified owner) –
Good quality.
Abhay Sharma (verified owner) –
Good quality.
Rahul Sharma (verified owner) –
Perfect hai bhai
Hariram (verified owner) –
Thankyou mynoteswala…. it helped me a lot.
Munna Yadav (verified owner) –
Good quality.
Payel Joshi (verified owner) –
Very Helpful
Vijay Malawat (verified owner) –
Good for revision .. ☺
Rohit kumar (verified owner) –
Super se bhi upar wale notes….☺☺☺
Akash Sharma (verified owner) –
Good quality.
Neelam Pandey (verified owner) –
Good quality.
Parul Sharma (verified owner) –
Very Helpful
Pakaj Jain (verified owner) –
Wonderful 👍
Munna Yadav (verified owner) –
Good quality.
Hariram (verified owner) –
It helps
Nikita Sharma (verified owner) –
Good quality Notes
Ritu singh (verified owner) –
Loved it
Jaswant Shekhawat (verified owner) –
Great notes… buy again
Pawan Agarwal (verified owner) –
Great notes… buy again
Tejinder singh (verified owner) –
Good quality.
Gungun Sharma (verified owner) –
Good quality.
Mukesh Bansal (verified owner) –
Good quality.
Hansika Khanna (verified owner) –
Good quality.
Shubham kurade (verified owner) –
Good quality.
Rudra Pratap Singh (verified owner) –
Good quality.
Ritu singh (verified owner) –
It helps
Sunita kumawat (verified owner) –
Super se bhi upar wale notes….☺☺☺
Riya kumari (verified owner) –
Told my friend about these notes
Neha Savita (verified owner) –
Good quality.
Komal Soni (verified owner) –
Concept clearing notes
Purva Soni (verified owner) –
Good quality.
Lokesh Khadiya (verified owner) –
Good quality.
Ritu singh (verified owner) –
Super se bhi upar wale notes….☺☺☺
Kiranjot Kaur (verified owner) –
Good quality.
Nihit Naithani (verified owner) –
Good quality.
Vimu Saini (verified owner) –
Good quality.
Abhishek Sharma (verified owner) –
Good quality.
Rupali Roy (verified owner) –
Good quality.
Depali Sharma (verified owner) –
Good quality.
Indresh Kumar (verified owner) –
Good quality.
Vishal Yadav (verified owner) –
It helps
Priya Joshi (verified owner) –
Told my friend about these notes
Sandhya Verma (verified owner) –
Good quality.
Anoushka gupta (verified owner) –
Loved it
Nihit Naithani (verified owner) –
Good quality.
Kusum Meena (verified owner) –
Helpful
Indu kumari (verified owner) –
Notes + Youtube video ka tadka … Maja aagya
Kuldeep Singh (verified owner) –
Good one
Sandhya Sani (verified owner) –
Good quality.
Vishal Jangid (verified owner) –
Good quality.
Anuj Sharma (verified owner) –
Good quality.
Khushi Saxena (verified owner) –
Good quality.
Ravi Sharma (verified owner) –
Nice one
Sarita Sharma (verified owner) –
Good quality.
Kusum Meena (verified owner) –
Told my friend about these notes
Indu kumari (verified owner) –
Perfect hai bhai
Shrikant (verified owner) –
Low price …. High return
Ruby Singh (verified owner) –
Good quality.
Harsh Kumawat (verified owner) –
Nice one
Komal Soni (verified owner) –
Awesome Notes
Riya kumari (verified owner) –
Good quality.
Vishal Jangid (verified owner) –
Good quality.
Suraj Jaiswal (verified owner) –
Good quality.
Monty singh (verified owner) –
Simple notes but powerful
Radhika Sherawat (verified owner) –
Good quality.
Sneha Saxena (verified owner) –
Good quality.
Vishal Yadav (verified owner) –
Excellent for students
Om Prakash Gupta (verified owner) –
Wonderful 👍
Yogesh Dakar (verified owner) –
Good quality.
Joy Tahazra (verified owner) –
Good quality.
Pramod Sharma (verified owner) –
Good quality.
Kanupriya Sharma (verified owner) –
Good quality.
Kundan Kumar (verified owner) –
Good quality.
Lokesh Gupta (verified owner) –
Good quality.
Rahul Sharma (verified owner) –
Awesome notes
Usha Saini (verified owner) –
Good quality.
Dinesh sharma (verified owner) –
Awesome Notes
Niharika Jain (verified owner) –
Good quality.
Kundan Jha (verified owner) –
Good quality.
Golu Singh (verified owner) –
Good quality.
Joy Tahazra (verified owner) –
Good quality.
Ruby Singh (verified owner) –
Good quality.
Aashu kumar (verified owner) –
Good quality.
Gudiya Gupta (verified owner) –
Good quality.
Rajiv Bhattacharjee (verified owner) –
Good quality.
Kusum Meena (verified owner) –
Best Notes…
Rita Gupta (verified owner) –
Good quality.
Monu Yadav (verified owner) –
Good quality.
Sajiya Khan (verified owner) –
Good quality.
Komal Soni (verified owner) –
Good quality Notes
Mukesh Kumar (verified owner) –
Good quality.
Lokesh Beniwal (verified owner) –
Concept clearing notes
Yogesh Meena (verified owner) –
GREAT Notes
Kavita Sharma (verified owner) –
Good quality.
Tejinder singh (verified owner) –
Good quality.
Dinesh sharma (verified owner) –
Bahut Acche notes hai
Pawan Agarwal (verified owner) –
Wonderful 👍
Mayank Kumar (verified owner) –
Good quality.
Vivek Singh Rawat (verified owner) –
Good quality.
Nihit Naithani (verified owner) –
Good quality.
Bitu roy (verified owner) –
Good quality.
Vaidehi (verified owner) –
Good quality.
Khushi Saxena (verified owner) –
Good quality.
Aman Bhardwaj (verified owner) –
Good quality.
Radhika Sherawat (verified owner) –
Good quality.
Rita Saxena (verified owner) –
Good quality.
Harsh Kumawat (verified owner) –
Perfect hai bhai
Aarti Kumari (verified owner) –
Perfect hai bhai
Komal Rathod (verified owner) –
Loved it
Khushi Saxena (verified owner) –
Good quality.
Chetana Saini (verified owner) –
Good quality.
Nikita Sharma (verified owner) –
Thankyou so much
Golu Singh (verified owner) –
Good quality.
Nikita Sharma (verified owner) –
Low price …. High return
Vijay Daswani (verified owner) –
Awesome Notes
Rakhi Saini (verified owner) –
Great notes… buy again
Vishal Jangid (verified owner) –
Only 29/- wow
Vijay Malawat (verified owner) –
It helps
Vijay Rathore (verified owner) –
Good quality.
Vijay Agarwal (verified owner) –
Loved it
Santosh Singh (verified owner) –
Good quality.
Anupam Sharma (verified owner) –
Good quality.
Jignash Patel (verified owner) –
Good quality.
Harsh Kumawat (verified owner) –
Nice notes + tricks
Suman pal (verified owner) –
Good quality.
Vijay Kumawat (verified owner) –
Awesome notes
Payel Joshi (verified owner) –
Thankyou mynoteswala…. it helped me a lot.
Pankaj Gupta (verified owner) –
It helps
Rita Gupta (verified owner) –
Good quality.
Jignash Patel (verified owner) –
Good quality.
Neha Yadav (verified owner) –
Awesome notes
Harsh Kumawat (verified owner) –
Simple notes but powerful
Depali Sharma (verified owner) –
Good quality.
Karnika Rathore (verified owner) –
Good quality.
Vivek Singh Rawat (verified owner) –
Good quality.
Madhulika Hirwar (verified owner) –
Good quality.
Hansika Khanna (verified owner) –
Good quality.
Nikita Sharma (verified owner) –
Good for revision .. ☺
Rohit kumar (verified owner) –
Good quality.
Usha Saini (verified owner) –
Good quality.
Khushi Saxena (verified owner) –
Good quality.
Vishal Jangid (verified owner) –
Concept was well explained.
Pawan Agarwal (verified owner) –
Nice notes + tricks
Babita Ghosh (verified owner) –
Good quality.
Kusum Meena (verified owner) –
Excellent for students
Anupam Sharma (verified owner) –
Good quality.
Shubham kurade (verified owner) –
Good quality.
Karnika Rathore (verified owner) –
Good quality.
Hariram (verified owner) –
Nice notes + tricks
Lokesh Beniwal (verified owner) –
Thankyou mynoteswala…. it helped me a lot.
Suman pal (verified owner) –
Good quality.
Vijay Kumawat (verified owner) –
I found it helpful 👍
Priya Joshi (verified owner) –
Perfect notes
Kaushal Singh (verified owner) –
Good quality.
Rajendra Sharma (verified owner) –
Good quality.
Rohit kumar (verified owner) –
Good quality.
Komal Rathod (verified owner) –
Great notes… buy again
Harsh Kumawat (verified owner) –
Good for revision .. ☺
Sanjana Shrimal (verified owner) –
Loved it
Kavita Soni (verified owner) –
The Best
Harsh Kumawat (verified owner) –
Simple explained
Nitya Singh (verified owner) –
Loved it…. Buy again
Surbhi Sati (verified owner) –
Good quality.
Kavita Sharma (verified owner) –
GREAT Notes
Shivam kumar Gupta (verified owner) –
Good quality.
Sabita Kumari (verified owner) –
Good quality.
Riya kumari (verified owner) –
Loved it…. Buy again
Niharika Jain (verified owner) –
Best Notes…
Rajiv Bhattacharjee (verified owner) –
Good quality.
Vipin Singh (verified owner) –
Good quality.
Sandhya Sharma (verified owner) –
Good quality.
Gudiya Gupta (verified owner) –
Good quality.
Anuj Sharma (verified owner) –
Good quality.
Vijay Daswani (verified owner) –
Told my friend about these notes
Payel Joshi (verified owner) –
Good quality.
Shiva Singh (verified owner) –
Good quality.
Indu kumari (verified owner) –
Nice notes + tricks
Depali Sharma (verified owner) –
Good quality.
Mohit Saxena (verified owner) –
GREAT Notes
Rupali Roy (verified owner) –
Good quality.
Rohit kumar (verified owner) –
Good quality Notes
Sandhya Sharma (verified owner) –
Good quality.
Nihit Naithani (verified owner) –
Good quality.
Kusum Meena (verified owner) –
The Best
Sonu Saini (verified owner) –
Good quality.
Suraj Jaiswal (verified owner) –
Good quality.
Rohit kumar (verified owner) –
GREAT Notes
Nikhil Mehra (verified owner) –
Good quality.
Babita Ghosh (verified owner) –
Good quality.
Manya Gupta (verified owner) –
Good quality.
Mohit Saxena (verified owner) –
Good one… 👍
Aarti Kumari (verified owner) –
GREAT Notes
Harish Rathore (verified owner) –
Good quality.
Harsh Kumawat (verified owner) –
Good quality.
Pankaj Gupta (verified owner) –
Good quality.
Mohit Saxena (verified owner) –
It helps
Pawan Agarwal (verified owner) –
Perfect notes
Kunal Singh (verified owner) –
Good quality.
Hansraj Sharma (verified owner) –
Good quality.
Jignash Patel (verified owner) –
Good quality.
Manisha Sharma (verified owner) –
Good quality.
Sarita Chaube (verified owner) –
Good quality.
Sandhya Yadav (verified owner) –
Good quality.
Yogesh Dakar (verified owner) –
Good quality.
Kavita Sharma (verified owner) –
Awesome notes
Sandhya Sharma (verified owner) –
Good quality.
Sabita Kumari (verified owner) –
Good quality.
Vaidehi (verified owner) –
Good quality.
Pramod Sharma (verified owner) –
Good quality.
Shivam Chaudhary (verified owner) –
Good quality.
Kunal Singh (verified owner) –
Good quality.
Rajiv Bhattacharjee (verified owner) –
Good quality.
Tejinder singh (verified owner) –
Good quality.
Kanupriya Sharma (verified owner) –
Good quality.
Karnika Goswami (verified owner) –
Good quality.
Gudiya Gupta (verified owner) –
Good quality.
Ritu singh (verified owner) –
Good quality.
Vijay Daswani (verified owner) –
Nice Notes…. Very Helpful
Aarti Kumari (verified owner) –
Good quality.
Sabita Sharma (verified owner) –
Good quality.
Karnika Goswami (verified owner) –
Good quality.
Jaswant Shekhawat (verified owner) –
Simple explained
Niharika Jain (verified owner) –
Bahut Acche notes hai
Depali Sharma (verified owner) –
Good quality.
Babita Ghosh (verified owner) –
Good quality.
Vijay Daswani (verified owner) –
Helpful
Hariram (verified owner) –
Simple notes but powerful
Rohit kumar (verified owner) –
Loved it…. Buy again
Abhishek Sharma (verified owner) –
Good quality.
Lakshmi Rawat (verified owner) –
Good quality.
Sabita Kumari (verified owner) –
Good quality.
Trisha Gupta (verified owner) –
Perfect notes
Priya Joshi (verified owner) –
Excellent for students
Anshra Kumari (verified owner) –
Good quality.
Dinesh Sharma (verified owner) –
Good quality.
Jaswant Shekhawat (verified owner) –
Recommended
Hansika Khanna (verified owner) –
Good quality.
Kavita Sharma (verified owner) –
Super se bhi upar wale notes….☺☺☺
Vijay Malawat (verified owner) –
Recommended
Rajiv Bhattacharjee (verified owner) –
Good quality.
Shivam Chaudhary (verified owner) –
Good quality.
Kusum Meena (verified owner) –
Super se bhi upar wale notes….☺☺☺
Aarti Kumari (verified owner) –
Nice Notes…. Very Helpful
Santosh Singh (verified owner) –
Good quality.
Rajini Rathore (verified owner) –
Good quality.
Sunita kumawat (verified owner) –
Simple notes but powerful
Lokesh Beniwal (verified owner) –
Concept was well explained.
Kaushal Singh (verified owner) –
Good quality.
Harsh Kumawat (verified owner) –
Nice one
Prachi Kumari (verified owner) –
Good quality.
Amit raj (verified owner) –
Good quality.
Sarita Chaube (verified owner) –
Good quality.
Gungun Sharma (verified owner) –
Good quality.
Vishal Yadav (verified owner) –
Nice notes + tricks
Aman Bhardwaj (verified owner) –
Good quality.
Pramod Soni (verified owner) –
Good quality.
Santosh Singh (verified owner) –
Good quality.
Kuldeep Kumar (verified owner) –
Good quality.
Lakshmi Rawat (verified owner) –
Good quality.
Jaswant Shekhawat (verified owner) –
Simple notes but powerful
Anoushka gupta (verified owner) –
Awesome Notes
Lokesh Beniwal (verified owner) –
The Best
Suraj Jaiswal (verified owner) –
Good quality.
Nikhil Yadav (verified owner) –
Very Helpful
Himanshu Gupta (verified owner) –
Good quality.
Kanupriya Sharma (verified owner) –
Good quality.
Rahul Sharma (verified owner) –
Super se bhi upar wale notes….☺☺☺
Vivek Verma (verified owner) –
Good quality.
Shivam Chaudhary (verified owner) –
Good quality.
Amit raj (verified owner) –
Good quality.
Prachi Pandey (verified owner) –
Good quality.
Saloni Singh (verified owner) –
Good quality.
Riya kumari (verified owner) –
Notes + Youtube video ka tadka … Maja aagya
Priya Joshi (verified owner) –
Simple explained
Pankaj Gupta (verified owner) –
Good quality.
Kundan Jha (verified owner) –
Good quality.
Deepa Shukla (verified owner) –
Good quality.
Sajiya Khan (verified owner) –
Good quality.
Sonali Kothwal (verified owner) –
Good quality.
Vishal Jangid (verified owner) –
Wonderful 👍
Surbhi Sati (verified owner) –
Good quality.
Anchal kumari (verified owner) –
It helps
Anchal kumari (verified owner) –
Helpful
Rajendra Joshi (verified owner) –
Good quality.
Kavita Sharma (verified owner) –
Awesome Notes
Tashi Gurjar (verified owner) –
Thankyou so much
Vijay Agarwal (verified owner) –
The Best
Mamta roy (verified owner) –
Good quality.
Indu kumari (verified owner) –
Loved it…. Buy again
Amit Ghosh (verified owner) –
Good quality.
Priya Joshi (verified owner) –
Good for revision .. ☺
Karnika Rathore (verified owner) –
Good quality.
Lokesh Saini (verified owner) –
Good quality.
Komal Soni (verified owner) –
Perfect hai bhai
Kavita Sharma (verified owner) –
Good quality.
Payel Joshi (verified owner) –
Helpful
Akash Sharma (verified owner) –
Good quality.
Rahul Sharma (verified owner) –
Thankyou so much
Nikhil Yadav (verified owner) –
Concept clearing notes
Lakshmi Rawat (verified owner) –
Good quality.
Kanupriya Sharma (verified owner) –
Good quality.
Vijay Daswani (verified owner) –
Thankyou mynoteswala…. it helped me a lot.
Payel Joshi (verified owner) –
Good quality.
Satyam Pandey (verified owner) –
Good quality.
Lokesh Beniwal (verified owner) –
Thankyou so much
Mukesh Bansal (verified owner) –
Good quality.
Swati Jain (verified owner) –
Good quality.
Manish Vaswani (verified owner) –
Loved it…. Buy again
Vijay Rathore (verified owner) –
Good quality.
Rakhi Saini (verified owner) –
Bahut Acche notes hai
Jitendra Maurya (verified owner) –
Good quality.
Radhika Sherawat (verified owner) –
Good quality.
Shivam Chaudhary (verified owner) –
Good quality.
Yogendra Tiwari (verified owner) –
Good quality.
Rehan Choudhary (verified owner) –
Good quality.
Vipin Singh (verified owner) –
Good quality.
Trisha Gupta (verified owner) –
Very affordable
Kusum Meena (verified owner) –
Chemistry made simple… go for it.
Sarita Chaube (verified owner) –
Good quality.
Sneha Saxena (verified owner) –
Good quality.
Nikhil Yadav (verified owner) –
Loved it
Kavita Sharma (verified owner) –
Nice notes + tricks
Udai Singh (verified owner) –
Good quality.
Mayank Choudhary (verified owner) –
Good quality.
Babita Ghosh (verified owner) –
Good quality.
Payel Joshi (verified owner) –
Good quality.
Shubham kurade (verified owner) –
Good quality.
Raunak Sharma (verified owner) –
Good quality.
Anshra Kumari (verified owner) –
Good quality.
Mohit Saxena (verified owner) –
Simple explained
Swati Sharma (verified owner) –
Good quality.
Kunal Singh (verified owner) –
Good quality.
Ravi Sharma (verified owner) –
Best Notes…
Mohit Gujar (verified owner) –
Good for revision .. ☺
Jignash Patel (verified owner) –
Good quality.
Vijay Kumawat (verified owner) –
Thankyou so much
Vishal Jangid (verified owner) –
Simple explained
Munna Yadav (verified owner) –
Good quality.
Usha Saini (verified owner) –
Good quality.
Mukesh Rajpurohit (verified owner) –
Good quality.
Rupendra Yadav (verified owner) –
Good quality.
Rita Saxena (verified owner) –
Good quality.
Rajendra Joshi (verified owner) –
Good quality.
Deepa Shukla (verified owner) –
Good quality.
Manoj Kesharwani (verified owner) –
Good quality.
Rajiv Bhattacharjee (verified owner) –
Good quality.
Harsh Kumawat (verified owner) –
Good quality.
Rajiv Bhattacharjee (verified owner) –
Good quality.
Sabita Sharma (verified owner) –
Good quality.
Sk Dhamani (verified owner) –
Good quality.
Riya kumari (verified owner) –
Good quality.
Rajendra Joshi (verified owner) –
Good quality.
Udai Singh (verified owner) –
Good quality.
Swati Sinha (verified owner) –
Good quality.
Rita Saxena (verified owner) –
Good quality.
Harshit Kumar (verified owner) –
Good quality.
Shubham kurade (verified owner) –
Good quality.
Lokesh Sharma (verified owner) –
Good quality.
Babita Ghosh (verified owner) –
Good quality.
Suman pal (verified owner) –
Good quality.
Udai Singh (verified owner) –
Good quality.
Sandhya Sani (verified owner) –
Good quality.
Manisha Sharma (verified owner) –
Good quality.
Rajendra Sharma (verified owner) –
Good quality.
Babita Ghosh (verified owner) –
Good quality.
Himanshu Upadhyay (verified owner) –
Good quality.
Tejinder singh (verified owner) –
Good quality.
Riya kumari (verified owner) –
Good quality.
Swati Jain (verified owner) –
Good quality.
Subhash Shahu (verified owner) –
Good quality.
getmynoteswala (verified owner) –
Good quality.